Authors: Gerald L. Klein, MD; Patrick Loebs; Freddy Byrth; Lee Schacter, PhD, MD; Michael Fath, PhD
Introduction
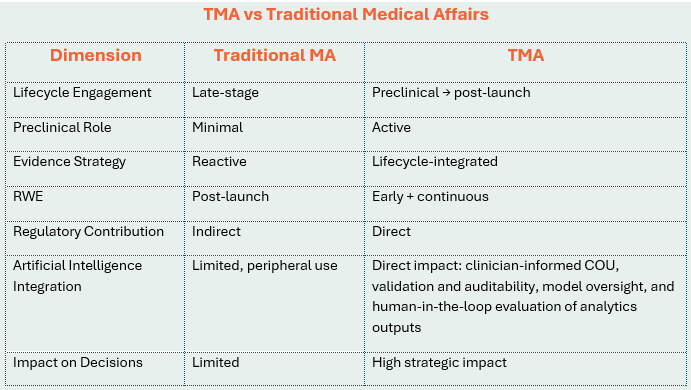
Medical Affairs (MA) is undergoing a fundamental transformation. Historically positioned as a late-stage, support-oriented function focused on scientific exchange and data dissemination, MA is now expected to play a far more strategic role in evidence generation and decision-making.
The drivers are clear:
● Increasing regulatory complexity
● Growing reliance on real-world evidence (RWE)
● Heightened expectations from payers and Health Technology Assessment ( HTA) bodies
● Persistent evidence gaps at launch
● Rapid integration of AI and advanced analytics
Traditional MA models—largely reactive and downstream—are no longer sufficient. A new model is emerging: Translational Medical Affairs (TMA).
What Is Translational Medical Affairs?
Unlike traditional MA inclusion strategies, in which MA is engaged late in the development cycle, TMA refers to a clinical development operating model where MA is embedded from the earliest stages of development as an integrated, lifecycle-spanning part of the interdisciplinary development team that augments the connections across preclinical development, clinical trial design and execution, real-world evidence generation, market access and payer strategy.
Why Preclinical Integration Is Critical
The most important decisions in drug development occur before the first patient is enrolled, including mechanism of action (MoA) selection, translational biomarker strategy, patient population definition, and early safety characterization.
Another critical consideration, particularly in therapeutic areas with a high density of active development programs, is the availability of patients for clinical trials. This includes not only the size of the addressable patient population, but also the number of ongoing and anticipated studies that may compete for the same patients, potentially impacting feasibility, enrollment timelines, and overall trial success.
In addition, early clinician input is essential in shaping study design and protocol execution, including inclusion and exclusion criteria and overall operational feasibility in real-world clinical settings.
TMA embeds Medical Affairs early, enabling validation of clinical relevance, alignment with ICH E8(R1) fit-for-purpose study design, anticipation of payer evidence needs, and identification of evidence gaps before costly Phase 1/2 trials.
Regulatory Alignment: A Key Driver
TMA aligns with evolving global regulatory expectations:
● ICH E6(R3) – Quality by Design and proactive risk management
● ICH E8(R1) – Fit-for-purpose study design aligned with stakeholder needs
● FDA Real-World Evidence Framework
● FDA Project Optimus
● FDA Artificial Intelligence/Machine Learning (AI/ML) guidance (January 2025
● EMA Artificial Intelligence Workplan (2023–2028)
These frameworks reinforce that evidence must be prospectively aligned with decisions—not retrospectively adapted.
Operationalizing Translational Medical Affairs
● Embed MA in preclinical development (Target Product Profile (TPP), biomarkers, MoA relevance)
The target product profile should be tightly linked to the overall clinical development plan, with periodic cross-functional clinical review to ensure continued alignment with emerging data, regulatory expectations, and strategic objectives.
● Develop a lifecycle evidence plan (regulatory, clinical, payer, RWE)
This lifecycle evidence plan should not function as a standalone document, but rather as an integrated component of the broader Clinical Development Plan, ensuring that evidence generation is coordinated, continuously updated, and aligned with key development and decision milestones.
● Integrate real-world evidence early (feasibility, endpoints, external validity)
Evolve MSLs into insight generators and evidence strategists
Strategic Value of TMA
● Higher probability of technical and regulatory success (PTRS)
● Reduced late-stage development risk
● Stronger payer positioning
● Faster clinical adoption
● More efficient resource utilization
● Generation of clinically meaningful insights to inform early prediction of risk-benefit profiles and logistical burden for both patients and clinical practice settings, including feasibility within routine care workflows.
The TMA Lifecycle Model
Preclinical → Clinical Development → Real-World Evidence → Market Access → Feedback to Development (continuous loop with bidirectional insight flow).
Conclusion
Translational Medical Affairs is a redefinition of the usual Medical Affairs function/construct. By integrating preclinical insight, clinical development, and real-world evidence, and the integration of artificial intelligence models and model development, TMA positions Medical Affairs as a strategic driver of development success, regulatory alignment, and clinical impact.
References
1. International Council for Harmonisation (ICH). E6(R3) Good Clinical Practice. 2025.
2. International Council for Harmonisation (ICH). E8(R1) General Considerations for Clinical Studies. 2021.
3. U.S. Food and Drug Administration. Framework for FDA’s Real-World Evidence Program. 2018.
4. U.S. Food and Drug Administration. Project Optimus. 2023.
5. European Medicines Agency. Artificial Intelligence Workplan 2023–2028.
6. U.S. Food and Drug Administration. Artificial Intelligence/Machine Learning (AI/ML)-Enabled Device Software Functions: Lifecycle Management and Marketing Submission Recommendations. Published January 2025.
7. U.S. Food and Drug Administration, Health Canada, Medicines and Healthcare products Regulatory Agency. Good Machine Learning Practice for Medical Device Development: Guiding Principles. Published 2021.
8. Sherman RE, Anderson SA, Dal Pan GJ, et al. Real-world evidence—what is it and what can it tell us? N Engl J Med. 2016;375(23):2293-2297.
9. Wong CH, Siah KW, Lo AW. Estimation of clinical trial success rates and related parameters. Biostatistics. 2019;20(2):273-286.
10. Drummond MF, Schwartz JS, Jönsson B, et al. Key principles for the improved conduct of health technology assessments for resource allocation decisions. Nat Rev Drug Discov. 2008;7(3):255-263.